Attention: If you want to read the book articles please send an email to g.helmchen@oci.uni-heidelberg.de to get access.
|
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 199. |
J. Melder, M. Heldner, J. Svetlicic, A. Adow, F. Rominger, M. Rudolph, G. Helmchen, A. S. K. Hashmi:
Photochemical Preparation of (Aza-)Indolines: Diastereoselective Synthesis and Polarity Reversal Strategy".
Adv. Synth. Catal. 2025, 367, e202500220.
|

|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 198. |
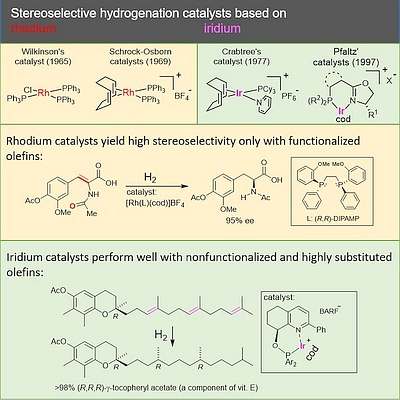
G. Helmchen:
Mechanistic Aspects of the Crabtree-Pfaltz Hydrogenation of Olefins – An Interplay of Experimentation and Quantum Chemical Computation".
Chem. Eur. J. 2023, 29, e202301488.
|
|
|
| 197. |
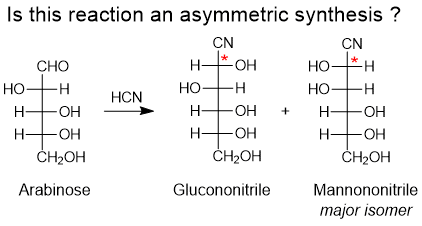
G. Helmchen:
Definition of the term asymmetric synthesis - History and revision".
Chirality 2023, 35, 1-8.
|
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 196. |
G. Helmchen:
"Rolf Huisgen".
in Heidelberger Akademie der Wissenschaften: Jahrbuch 2020. Paderborn, 2021, 126-129. |
|
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
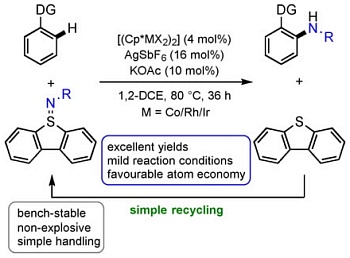
| 195. |
P. Antoni, A. Mackenroth, F. Mulks, M. Rudolph, G. Helmchen, A. S. K. Hashmi:
Dibenzothiophenesulfilimines: A Convenient Approach to Intermolecular Rhodium-Catalysed C-H Amidation".
Chem. Eur. J. 2020, 26, 8235-8238.
|
|
|
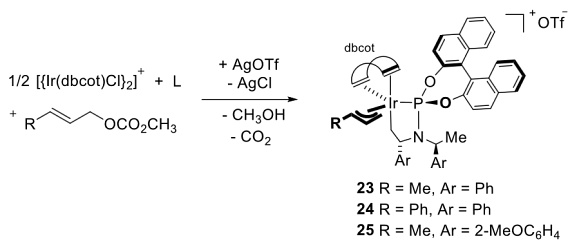
| 194. |
G. Helmchen:
Discussion Addendum for: "Dibenzo[a,e]cyclooctene: MultigramSynthesis of a Bidentate Ligand".
Organic Syntheses 2020, 97, 66-78.
|
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 193. |
J.-P. Qu, G. Helmchen, Z.-P. Yang, W. Zhang, S.-L. You:
"Iridium-Catalyzed, Enantioselective, Allylic Alkylations with Carbon Nucleophiles".
Organic Reactions 2019, 99, 423-632.
|
| |
|
| 192. |
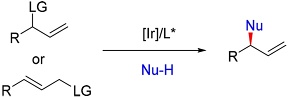
Q. Cheng, H.-F. Tu, C. Zheng, J.-P. Qu, G. Helmchen, S.-L. You:
"Iridium-Catalyzed Asymmetric Allylic Substitution Reactions".
Chem. Rev. 2019, 119, 1855-1969.
|
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
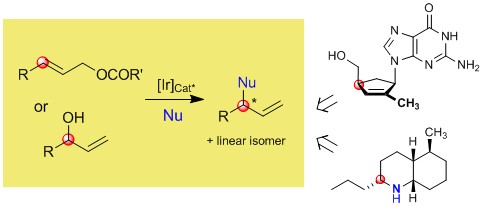
| 191. |
J. Qu, G. Helmchen:
"Applications of Iridium-Catalyzed Asymmetric Allylic Substitution Reactions in Target-Oriented Synthesis".
Acc. Chem. Res. 2017, 50, 2539-2555.
|
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 190. |
G. Helmchen:
"The 50th Anniversary of the Cahn–Ingold–Prelog Specification of Molecular Chirality".
Angew. Chem. Int. Ed. 2016, 55, 6798–6799.
"50 Jahre Spezifikation der molekularen Chiralität durch Cahn, Ingold und Prelog".
Angew. Chem. 2016, 128, 6910-6911.
|
| |
|
| 189. |
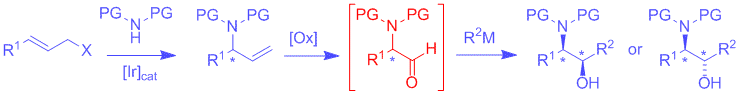
K. Seehafer, C. Malakar, M. Bender, J. Qu, C. Liang, G. Helmchen:
"Iridium-Catalyzed Asymmetric Allylic Substitutions with Bulky Amines/Oxidative Double Bond Cleavage – Entry into the Reetz Synthesis of Amino Alcohols".
Eur. J. Org. Chem. 2016, 493-501. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 188. |
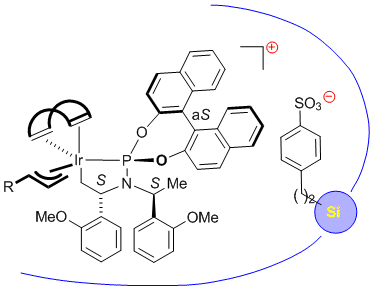
C. Malakar, G. Helmchen:
"Immobilized Catalysts for Iridium-Catalyzed Allylic Amination - Rate Enhancement by Immobilization".
Chem. Eur. J. 2015, 21 (19), 7127-7134. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
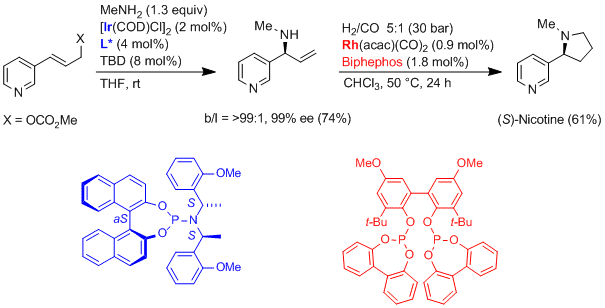
| 187. |
G. Helmchen:
"Ir-Catalyzed Asymmetric Allylic Substitution Reactions – Fundamentals and Applications in Natural Products synthesis".
in L. H. Gade, P. Hofmann: Molecular Catalysis. Wiley-VCH, Weinheim, 2014, 235-254. |
|
| |
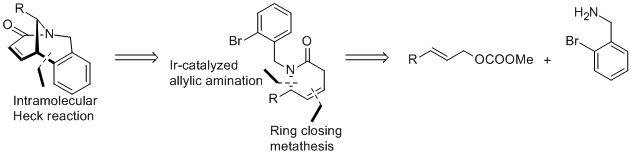
| 186. |
G. Satyanarayana, G. Helmchen:
"Enantioselective Syntheses of Bicyclic Lactams Based on Iridium-Catalyzed Asymmetric Allylic Substitution and Heck Cyclization".
Eur. J. Org. Chem. 2014, 2242–2252. |
|
| |
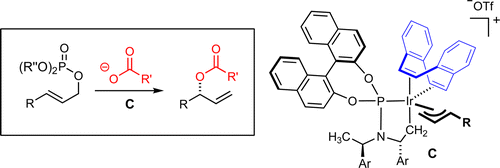
| 185. |
J. Qu, L. Roßberg, G. Helmchen:
"Enantio- and Regioselective Iridium-Catalyzed Allylic Esterification".
J. Am. Chem. Soc. 2014, 136 (4), 1272-1275. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
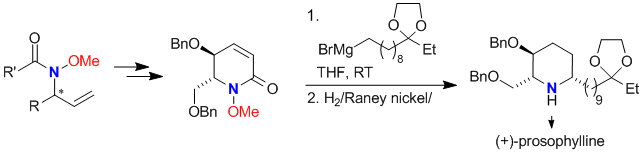
| 184. |
M. Jäkel, J. Qu, T. Schnitzer, G. Helmchen:
"Addition of Organometallic Reagents to Chiral N-Methoxylactams: Enantioselective Syntheses of Pyrrolidines and Piperidines".
Chem. Eur. J. 2013, 19, 16746–16755. |
|
| |
| 183. |
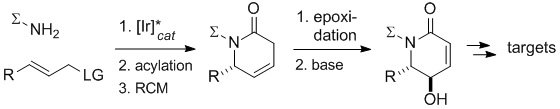
J. Hoecker, G. C. Rudolf, F. Bächle, S. Fleischer, B. D. Lindner, G. Helmchen:
"Enantio- and Diastereoselective Syntheses of 3-Hydroxypiperidines through Iridium-Catalyzed Allylic Substitution".
Eur. J. Org. Chem. 2013, 5149–5159. |
|
| |
| 182. |
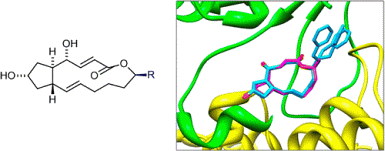
K. Seehafer, F. Rominger, G. Helmchen, M. Langhans, D. G. Robinson, B. özata, B. Brügger, J. R. P. M. Strating, F. J. M. van Kuppeveld, C. D. Klein:
"Synthesis and Biological Properties of Novel Brefeldin A Analogues".
J. Med. Chem. 2013, 56, 5872-5884. |
|
| |
| 181. |
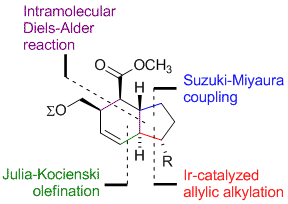
M. Gärtner, G. Satyanarayana, S. Förster, G. Helmchen:
"Syntheses of the Hexahydroindene Cores of Indanomycin and Stawamycin by Combinations of Iridium-Catalyzed Asymmetric Allylic Alkylations and Intramolecular Diels–Alder Reactions".
Chem. Eur. J. 2013, 18, 400–405. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 180. |
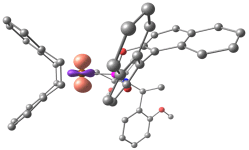
J. Raskatov, M. Jäkel, B. Straub, F. Rominger, G. Helmchen:
"Iridium-Catalyzed Allylic Substitutions with Cyclometalated Phosphoramidite Complexes Bearing a Dibenzocyclooctatetraene Ligand: Preparation of (p-Allyl)Ir Complexes and Computational and NMR Spectroscopic Studies".
Chem. Eur. J. 2012, 18, 14314–14328. |
|
| |
| 179. |
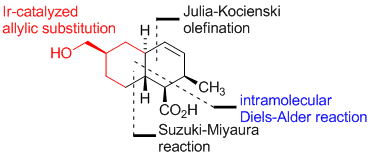
M. Gärtner, D. Kossler, D. Pflästerer, G. Helmchen:
"Enantioselective Total Synthesis and Absolute Configuration of Apiosporic Acid".
J. Org. Chem. 2012, 77, 4491–4495. |
|
| |
| 178. |
G. Franck, M. Brill, G. Helmchen:
"Dibenzo[a,e]cyclooctene: Multi-gram Synthesis of a Bidentate Ligand".
Organic Syntheses 2012, 89, 55-65. |
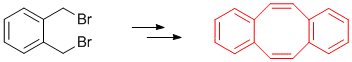
|
| |
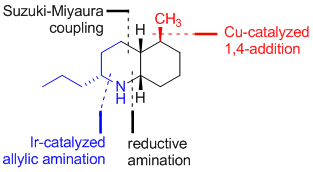
| 177. |
M. Gärtner, J. Qu, G. Helmchen:
"Enantioselective Syntheses of the Alkaloids cis-195A (Pumiliotoxin C) and trans-195A Based on Multiple Applications of Asymmetric Catalysis".
J. Org. Chem. 2012, 77, 1186-1190. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
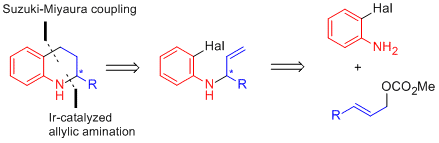
| 176. |
G. Satyanarayana, D. Pflästerer, G. Helmchen:
"Enantioselective Syntheses of Tetrahydroquinolines Based on Iridium-Catalyzed Allylic Substitutions: Total Syntheses of (+)-Angustureine and (–)-Cuspareine".
Eur. J. Org. Chem. 2011, 6877–6886. |
|
| |
| 175. |
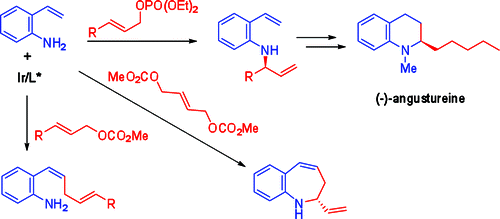
Ke-Yin Ye, Hu He, Wen-Bo Liu, Li-Xin Dai, Günter Helmchen, Shu-Li You:
"Iridium-Catalyzed Allylic Vinylation and Asymmetric Allylic Amination Reactions with o-Aminostyrenes".
J. Am. Chem. Soc. 2011, 133, 19006–19014. |
|
| |
| 174. |
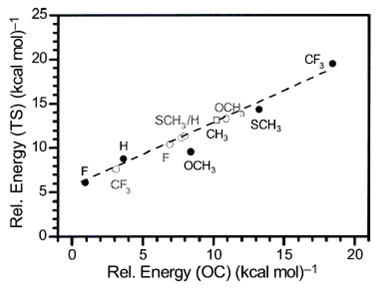
J. Raskatov, G. Helmchen:
" Effects of Substituents on the Regioselectivity of Palladium-Catalysed Allylic Substitutions - A DFT Study".
in P. Comba: Modeling of Molecular Properties. Wiley-VCH, Weinheim, 2011, p.191-206. |
|
| |
| 173. |
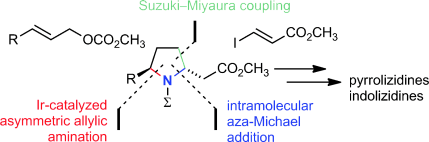
M. Gärtner, R. Weihofen, G. Helmchen:
"Enantioselective Syntheses of 2,5-Disubstituted Pyrrolidines Based on Iridium-Catalyzed Allylic Aminations - Total Syntheses of Alkaloids from Amphibian Skins".
Chem. Eur. J. 2011, 17, 7605–7622. |
|
| |
| 172. |
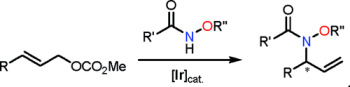
M. Gärtner, M. Jäkel, M. Achatz, Ch. Sonnenschein, O. Tverskoy, G Helmchen:
"Enantioselective Iridium-Catalyzed Allylic Substitutions with Hydroxamic Acid Derivatives as N-Nucleophiles".
Organic Letters 2011, 13(11), 2810–2813. |
|
| |
| 171. |
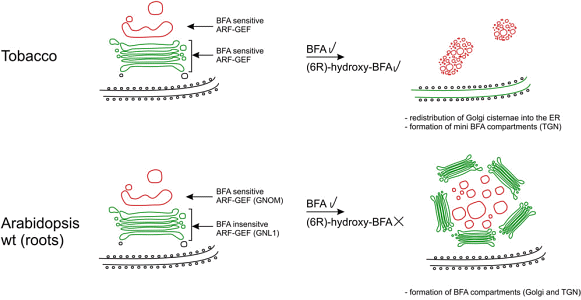
M. Langhans, S. Förster, D. G. Robinson, G. Helmchen:
"Differential effects of the brefeldin A analogue (6R)-hydroxy-BFA in tobacco and Arabidopsis".
Journal of Experimental Botany 2011, 62(8), 2949-2957. |
|
| |
| 170. |
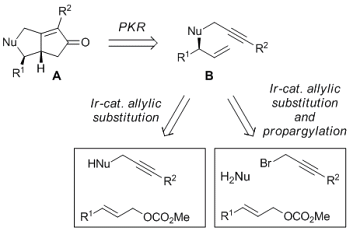
A. Farwick, J. Engelhart, O. Tverskoy, C. Welter, Q. Umlauf, F. Rominger, W. Kerr, G. Helmchen:
"Bicyclic Cyclopentenones via the Combination of an Iridium- Catalyzed Allylic Substitution with a Diastereoselective Intramolecular Pauson–Khand Reaction".
Adv. Synth. Catal. 2011, 353, 349–370. |
|
| |
| 169. |
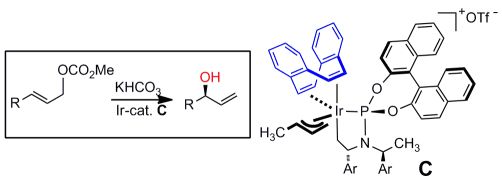
M. Gärtner, S. Mader, K. Seehafer, G. Helmchen:
"Enantio- and Regioselective Iridium-Catalyzed Allylic Hydroxylation".
J. Am. Chem. Soc. 2011, 133, 2072–2075. |
|
| |
| 168. |
S. Förster, E. Persch, O. Tverskoy, F. Rominger, G. Helmchen, C. Klein, B. Gönen, B. Brügger:
"Syntheses and Biological Properties of Brefeldin Analogues".
Eur. J. Org. Chem. 2011, 5, 878–891 |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 167. |
M. Schelwies, A. Farwick, F. Rominger, G. Helmchen:
"Platinum(II) Chloride-Catalyzed Stereoselective Domino Enyne Isomerization/Diels-Alder Reaction".
J. Org. Chem. 2010, 75, 7917–7919. |

|
| |
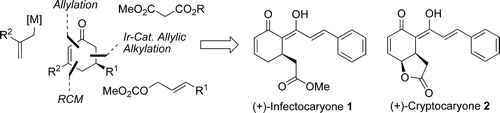
| 166. |
G. Franck, K. Brödner, G. Helmchen:
"Enantioselective Modular Synthesis of Cyclohexenones: Total Syntheses of (+)-Crypto- and (+)-Infectocaryone".
Organic Letters 2010, 12(17), 3886-3889. |
|
| |
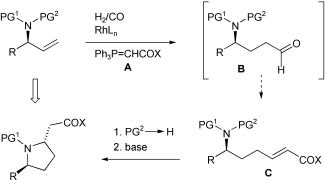
| 165. |
A. Farwick, G. Helmchen:
"Stereoselective Synthesis of ß-Proline Derivatives from Allylamines via Domino Hydroformylation/Wittig Olefination and Aza-Michael Addition".
Adv. Synth. Catal. 2010, 352, 1023–1032. |
|
| |
| 164. |
S. Förster, G. Helmchen, U. Kazmaier:
" Enantioselective Allylic Substitutions with Carbon Nucleophiles".
in I. Ojima: Catalytic Asymmetric Synthesis. 3rd ed. Wiley, New Jersey, 2010, p.497-641. |

|
| |
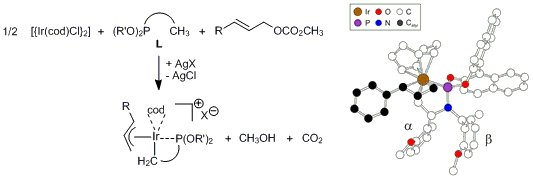
| 163. |
J. Raskatov, S. Spiess, C. Gnamm, K. Brödner, F. Rominger, G. Helmchen:
"Ir-Catalysed Asymmetric Allylic Substitutions with Cyclometalated (Phosphoramidite)Ir Complexes—Resting States, Catalytically Active (p-Allyl)Ir Complexes and Computational Exploration".
Chem. Eur. J. 2010, 22, 6601–6615.
see also: Supporting Information to the Article |
|
| |
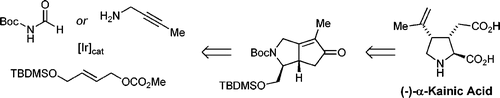
| 162. |
A. Farwick, G. Helmchen:
"Enantioselective Total Synthesis of (-)-a-Kainic Acid".
Organic Letters 2010, 12, 1108–1111. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
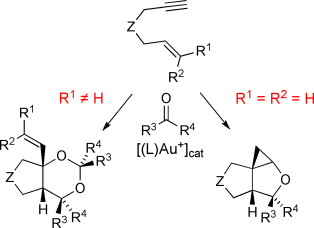
| 161. |
M. Schelwies, R. Moser, A. Dempwolff, F. Rominger, G. Helmchen:
"Gold-Catalyzed Intermolecular Addition of Carbonyl Compounds to 1,6-Enynes: Reactivity, Scope, and Mechanistic Aspects".
Chem. Eur. J. 2009, 15, 10888–10900. |
|
| |
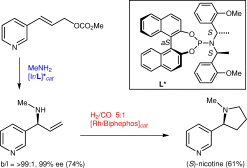
| 160. |
P. Dübon, A. Farwick, G. Helmchen:
"Enantioselective Syntheses of 2-Substituted Pyrrolidines from Allylamines by Domino Hydroformylation-Condensation: Short Syntheses of (S)-Nicotine and the Alkaloid 225C".
Synlett 2009, 9, 1413-1416. |
|
| |
| 159. |
S. Spiess, J. Raskatov, C. Gnamm, K. Brödner, G. Helmchen:
"Ir-Catalyzed Asymmetric Allylic Substitutions with (Phosphoramidite) Ir Complexes - Resting States, Synthesis, und Characterization of Catalytically Active (p-Allyl)Ir Complexes".
Chem. Eur. J. 2009, 15, 11087–11090. |
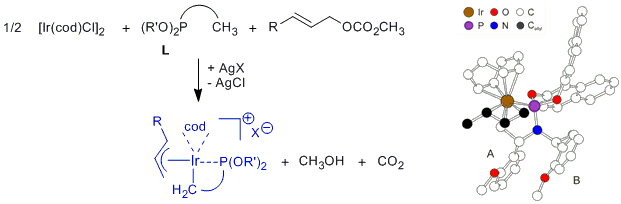
|
| |
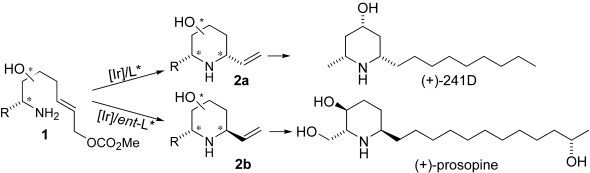
| 158. |
C. Gnamm, K. Brödner, C. Krauter, G. Helmchen:
"A Configurational Switch Based on Iridium-Catalyzed Allylic Cyclization: Application in Asymmetric Total Syntheses of Prosopis, Dendrobate, and Spruce Alkaloids".
Chem. Eur. J. 2009, 15, 10514–10532. |
|
| |
| 157. |
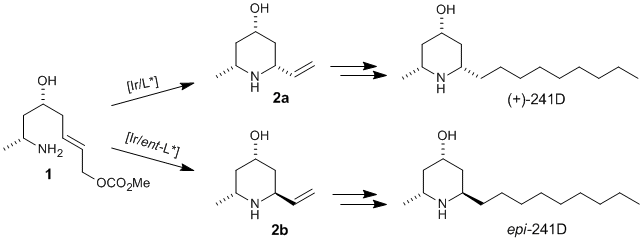
C. Gnamm, C. Krauter, K. Brödner, G. Helmchen:
"Stereoselective Synthesis of 2,6-Disubstituted Piperidines Using the Iridium-Catalyzed Allylic Cyclization as Configurational Switch: Asymmetric Total Synthesis of (+)-241D and Related Piperidine Alkaloids".
Chem. Eur. J. 2009, 15, 2050–2054. |
|
| |
| 156. |
G. Helmchen:
"Iridium-Catalyzed Asymmetric Allylic Substitutions".
in L. A. Oro, C. Claver: Iridium Complexes in Organic Synthesis. Wiley-VCH, Weinheim, 2009, p.211-250. |
| |
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 155. |
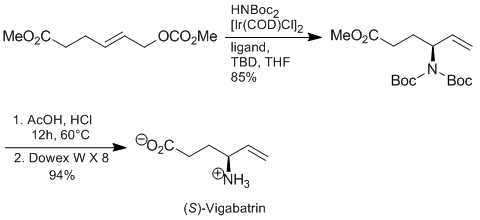
C. Gnamm, G. Franck, N. Miller, T. Stork, K. Brödner, G. Helmchen:
"Enantioselective Iridium-Catalyzed Allylic Aminations of Allylic Carbonates with Functionalized Side Chains. Asymmetric Total Synthesis of (S)-Vigabatrin".
Synthesis 2008, 20, 3331–3350. |
|
| |
| 154. |
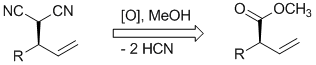
S. Förster, O. Tverskoy, G. Helmchen:
"Malononitrile as Acylanion Equivalent".
Synlett 2008, 18, 2803–2806. |
|
| |
| 153. |
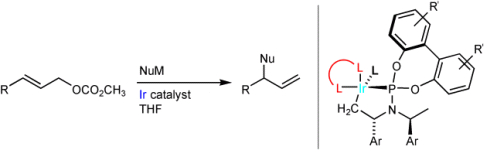
S. Spiess, C. Welter, G. Franck, J.-P. Taquet, G. Helmchen:
"Iridium-Catalyzed Asymmetric Allylic Substitutions - Very High Regioselectivity and Air Stability with a Catalyst Derived from Dibenzo[a,e]cyclooctatetraene and a Phosphoramidite".
Angew. Chem. Int. Ed. 2008, 47, 7652-7655.
"Iridiumkatalysierte asymmetrische allylische Substitutionen – sehr hohe Regioselektivität und Luftstabilität mit einem Katalysator auf der Basis von Dibenzo[a,e]cyclooctatetraen und einem Phosphoramidit".
Angew. Chem. 2008, 120, 7764-7767. |
|
| |
| 152. |
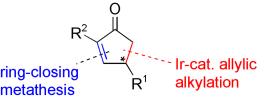
P. Dübon, M. Schelwies, G. Helmchen:
"Preparation of 2,4-Disubstituted Cyclopentenones by Enantioselective Iridium-Catalyzed Allylic Alkylation: Synthesis of 2’-Methylcarbovir and TEI-9826".
Chem. Eur. J. 2008, 14, 6722-6733. |
|
| |
| 151. |
S. Förster, G. Helmchen:
"Stereoselective Synthesis of a Lactam Analogue of Brefeldin C".
Synlett 2008, 6, 831-836. |
|
| |
| 150. |
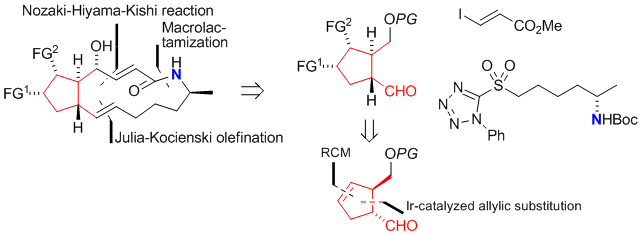
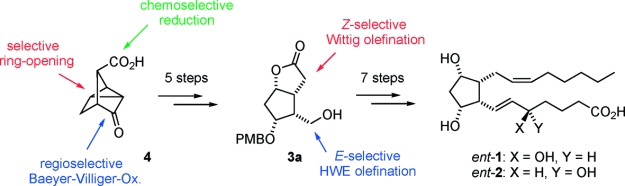
P. Elsner, P. Jetter, K. Brödner, G. Helmchen:
"Stereoselective Synthesis of a cis-1,2-Dialkylcyclopentane Building Block and Its Application in Isoprostane Synthesis (5-ent-F2c-IsoP)".
Eur. J. Org. Chem. 2008, 15, 2551–2563. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 149. |
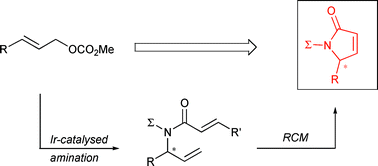
S. Spiess, C. Berthold, R. Weihofen, G. Helmchen:
"Synthesis of α,β-Unsaturated γ-Lactams via Asymmetric Iridium-Catalysed Allylic Substitution".
Org. Biomol. Chem. 2007, 5, 2357–2360. |
|
| |
| 148. |
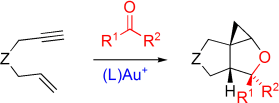
M. Schelwies, A. Dempwolff, F. Rominger, G. Helmchen:
"Gold-Catalyzed Intermolecular Addition of Carbonyl Compounds to 1,6-Enynes".
Angew. Chem. Int. Ed. 2007, 46, 5598–5601.
"Gold-katalysierte intermolekulare Addition von Carbonylverbindungen an 1,6-Enine".
Angew. Chem. 2007, 119, 5694–5697. |
|
| |
| 147. |
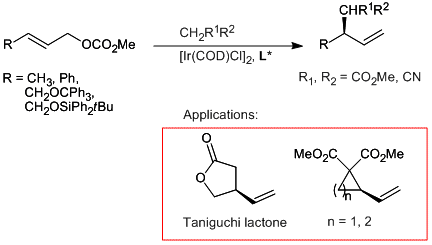
C. Gnamm, S. Förster, N. Miller, K. Brödner, G. Helmchen:
"Enantioselective Iridium-Catalyzed Allylic Alkylations – Improvements and Applications Based on Salt-Free Reaction Conditions".
Synlett 2007, 5, 790–794. |
|
| |
| 146. |
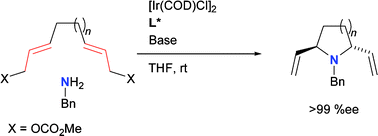
A. Dahnz, P. Dübon, M. Schelwies, R. Weihofen, G. Helmchen:
"Iridium-catalyzed asymmetric allylic substitutions".
Chem. Comm. 2007, 675–691. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 145. |
G. Helmchen:
"Asymmetric Allylic Substitutions".
in M. Christmann, S. Bräse (Eds.): Asymmetric Synthesis - The Essentials. Wiley-VCH, Weinheim, 2006, p.95-99. |
| |
| |
| 144. |
D. A. Evans, G. Helmchen, M. Rüping:
"Chiral Auxiliaries in Asymmetric Synthesis".
in M. Christmann, S. Bräse (Eds.): Asymmetric Synthesis - The Essentials. Wiley-VCH, Weinheim, 2006, p.3-9. |
| |
| |
| 143. |
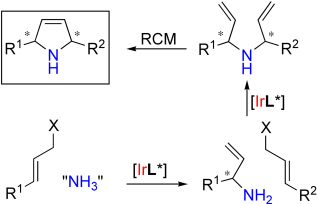
R. Weihofen, O. Tverskoy, G. Helmchen:
"Salt-Free Iridium-Catalyzed Asymmetric Allylic Aminations with N,N-Diacylamines and ortho-Nosylamide as Ammonia Equivalents".
Angew. Chem. Int. Ed. 2006, 45, 5546-5549
"Salzfreie Iridium-katalysierte allylische Aminierungen mit N,N-Diacylaminen und ortho-Nosylamid als Ammoniak-Äquivalenten".
Angew. Chem. 2006, 118, 5673-5676.
|
|
| |
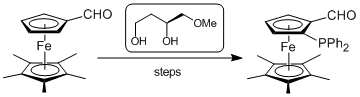
| 142. |
F. Geisler, G. Helmchen:
"A Straightforward Synthesis of (3S)-4-Methoxybutane-1,3-diol and its Use as Chiral Auxiliary for the Preparation of (pS)-1-(Diphenylphosphino)-2-formyl-1',2',3',4',5'-pentamethylferrocene".
Synthesis 2006, 13, 2201–2205. |
|
| |
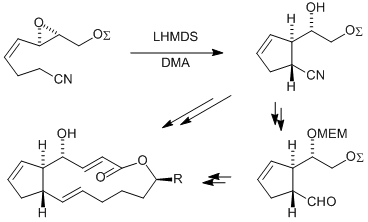
| 141. |
T. Hübscher, G. Helmchen:
"Enantioselective Formal Synthesis of Brefeldin A and Analogues via Anionic Cyclization of an Alkenyl Epoxide".
Synlett 2006, 9, 1323–1326. |
|
| |
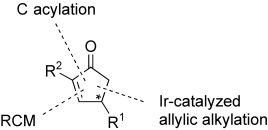
| 140. |
M. Schelwies, P. Dübon, G. Helmchen:
"Enantioselective Modular Synthesis of 2,4-Disubstituted Cyclopentenones by Iridium-Catalyzed Allylic Alkylation".
Angew. Chem. Int. Ed. 2006, 45, 2466-2469.
"Enantioselektive modulare Synthese von 2,4-disubstituierten Cyclopentenonen durch Iridium-katalysierte allylische Alkylierung".
Angew. Chem. 2006, 118, 2526-2529. |
|
| |
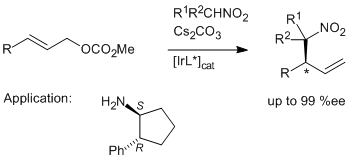
| 139. |
A. Dahnz, G. Helmchen:
"Iridium-Catalyzed Enantioselective Allylic Substitutions with Aliphatic Nitro Compounds as Prenucleophiles".
Synlett 2006, 5, 697-700. |
|
| |
| 138. |
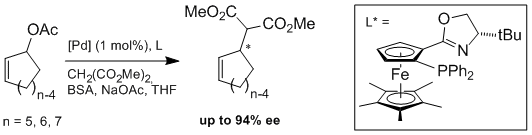
F. Geisler, G. Helmchen:
"Chiral Phosphinooxazolines with a Pentamethylferrocene Backbone - Synthesis and Use as Ligands in Asymmetric Catalysis".
J. Org. Chem. 2006, 71, 2486-2492. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 137. |
A. Stang, G. Helmchen:
"Enantioselective Syntheses and Fragrance Properties of the Four Stereoisomers of Magnolione® (Magnolia Ketone)".
Helv. Chim. Acta 2005, 88, 2738-2746.
|
| |
| 136. |
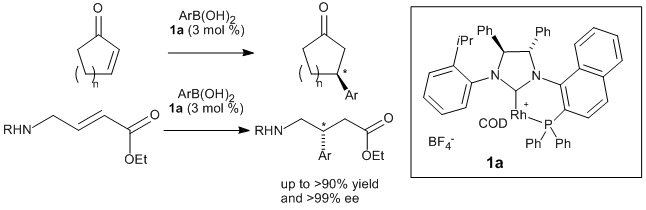
J.-M. Becht, E. Bappert, G. Helmchen:
"Application of Rhodium Complexes of Chiral Diphenylphosphino-Functionalized N-Heterocyclic Carbenes as Catalysts in Enantioselective Conjugate Additions of Arylboronic Acids".
Adv. Synth. Catal. 2005, 347, 1495–1498. |
|
| |
| 135. |
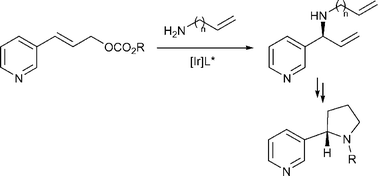
C. Welter, R. Moreno, S. Streiff, G. Helmchen:
"Enantioselective synthesis of (+)(R)- and (-)(S)-nicotine based on Ir-catalysed allylic amination".
Org. Biomol. Chem. 2005, 3, 3266–3268. |
|
| |
| 134. |
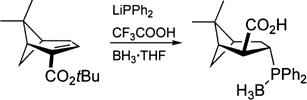
B. Wiese, G. Knühl, D. Flubacher, J. Prieß, B. Ulriksen, K. Brödner, G. Helmchen:
"Syntheses of New Chiral Phosphane Ligands by Diastereoselective Conjugate Addition of Phosphides to Enantiomerically Pure Acceptor-Substituted Olefins from the Chiral Pool".
Eur. J. Org. Chem. 2005, 3246–3262. |
|
| |
| 133. |
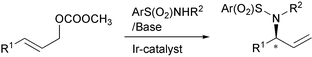
R. Weihofen, A. Dahnz, O. Tverskoy and G. Helmchen:
"Highly enantioselective iridium-catalysed allylic aminations with anionic N-nucleophiles".
Chem. Commun. 2005, 3541–3543. |
|
| |
| 132. |
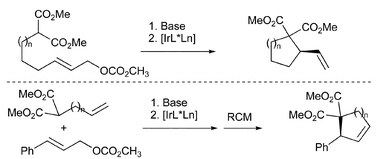
S. Streiff, C. Welter, M. Schelwies, G. Lipowsky, N. Miller and G. Helmchen:
"Carbocycles via enantioselective inter- and intramolecular iridium-catalysed allylic alkylations".
Chem. Commun. 2005, 2957–2959. |
|
| |
| 131. |
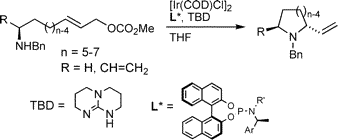
C. Welter, A. Dahnz, B. Brunner, S. Streiff, P. Dübon, G. Helmchen:
"Highly Enantioselective Syntheses of Heterocycles via Intramolecular Ir-Catalyzed Allylic Amination and Etherification".
Organic Letters 2005, 7, 1239-1242. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 130. |
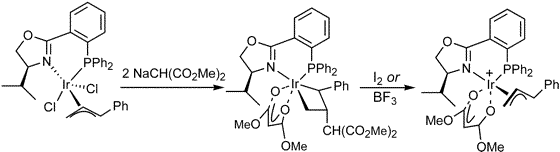
C. Garcia-Yebra, J. Janssen, F. Rominger, G. Helmchen:
"Asymmetric Iridium(I)-Catalyzed Allylic Alkylation of Monosubstituted Allylic Substrates with Phosphinooxazolines as Ligands. Isolation, Characterization, and Reactivity of Chiral (Allyl)iridium(III) Complexes".
Organometallics 2004, 23, 5459-5470. |
|
| |
| 129. |
G. Lipowsky, N. Miller, G. Helmchen:
"Regio- and Enantioselective Iridium- Catalyzed Allylic Alkylation with In Situ Activated P,C-Chelate Complexes".
Angew. Chem. Int. Ed. 2004, 43, 4595-4597.
"Regio- und enantioselektive Iridium-katalysierte allylische Alkylierung mit in situ aktivierten P,C-Chelatkomplexen".
Angew. Chem. 2004, 116, 4695-4698. |
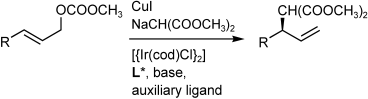
|
| |
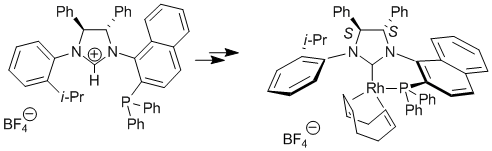
| 128. |
E.Bappert, G. Helmchen:
"Synthesis and Application of Complexes of a Novel Chiral Diphenylphosphino-Functionalized N-Heterocyclic Carbene".
Synlett 2004, 10, 1789–1793. |
|
| |
| 127. |
G. Paradies, M. Ernst, G. Helmchen:
"Application of Allylic Substitutions in Natural Products Synthesis".
Pure Appl. Chem. 2004, 76(3), 495-506. |
| |
| |
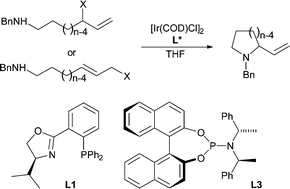
| 126. |
C. Welter, O. Koch, G. Lipowsky, G. Helmchen:
"First intramolecular enantioselective iridium-catalysed allylic aminations".
Chem. Commun. 2004, 896-897 |
|
| |
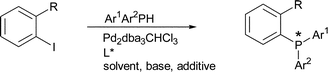
| 125. |
C. Korff, G. Helmchen:
"Preparation of chiral triarylphosphines by Pd-catalysed asymmetric P–C cross-coupling".
Chem. Commun. 2004, 530-531. |
|
| |
| 124. |
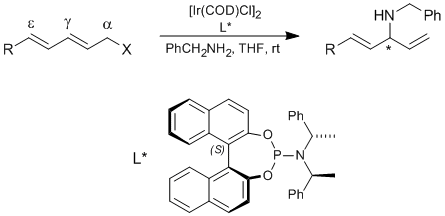
G. Lipowsky, G. Helmchen:
"Regio- and enantioselective iridium-catalysed allylic aminations and alkylations of dienyl esters".
Chem. Commun. 2004, 116-117. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 123. |
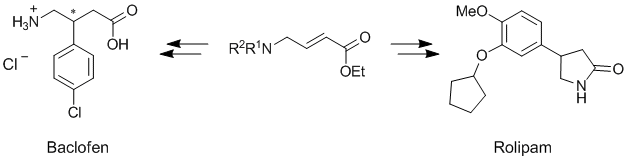
J.-M. Becht, O. Meyer, G. Helmchen:
"Enantioselective Synthesis of (-)-(R)-Rolipram, (-)-(R)-Baclofen and Other GABA Analogues via Rhodium-Catalyzed Conjugate Addition of Arylboronic Acids ".
Synthesis 2003, 18, 2805-2810. |
|
| |
| 122. |
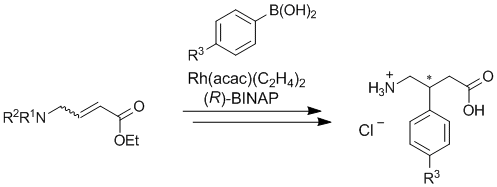
O. Meyer, J.-M. Becht, G. Helmchen:
"Enantioselective Synthesis of (-)-(R)-Baclofen and Analogues via Rhodium Catalysed Conjugate Addition of Boronic Acids".
Synlett 2003, 10, 1539-1541. |
|
| |
| 121. |
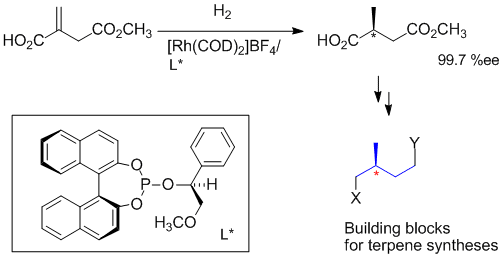
M. Ostermeier, B. Brunner, C. Korff, G. Helmchen:
"Highly Enantioselective Rhodium-Catalyzed Hydrogenation of 2-(2-Methoxy-2-oxoethyl)acrylic Acid - A Convenient Access of Enantiomerically Pure Isoprenoid Building Blocks".
Eur. J. Org. Chem. 2003, 3453-3459. |
|
| |
| 120. |
E. Montenegro, B. Gabler, G. Paradies, M. Seemann, G. Helmchen:
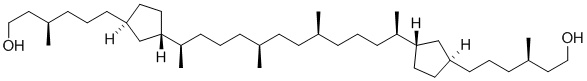
"Determination of the Configuration of an Archaea Membrane Lipid Containing Cyclopentane Rings by Total Synthesis".
Angew. Chem. Int. Ed. 2003, 42, 2419-2421.
"Bestimmung der Konfiguration eines Cyclopentanringe enthaltenden Archaea-Membranlipids durch Totalsynthese".
Angew. Chem. 2003 (2), 115, 2521-2523. |
|
| |
| 119. |
M. Seemann, M. Schöler, S. Kudis, G. Helmchen:
"Syntheses of Enantiomerically Pure Cyclopent-2-ene-1-carboxylic Acid and (Cyclopent-2-enyl)acetic Acid by Enantioselective Palladium-Catalyzed Allylic Alkylations - Synthesis of Enantiomerically Pure (-)-Chaulmoogric Acid".
Eur. J. Org. Chem. 2003 (11), 2122-2127.
|
| |
| 118. |
B. Bartels, C. Garcia-Yebra, G. Helmchen:
"Asymmetric IrI-Catalysed Allylic Alkylation Of Monosubstituted Allylic Acetates With Phosphorus Amidites As Ligands".
Eur. J. Org. Chem. 2003 (6), 1097-1103.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 117. |
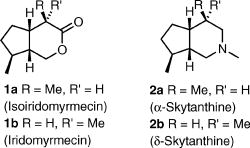
M. Ernst, G. Helmchen:
"A Novel Route to Iridoids: Enantioselective Synthesis of Isoiridomyrmecin and α-Skythanthine".
Synthesis 2002, 14, 1953-1955. |
|
| |
| 116. |
M. Ernst, G. Helmchen:
"A New Synthesis Route to Enantiomerically Pure Jasmonoids".
Angew. Chem. Int. Ed. 2002, 41(21), 4054-4056.
"Ein neuer Syntheseweg zu enantiomerenreinen Jasmonoiden".
Angew. Chem. 2002, 114, 4231-4234.
|
| |
| 115. |
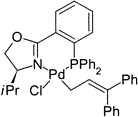
M. Kollmar, G. Helmchen:
"An (η1-Allyl)Pd Complex of a Chiral Bidentate Ligand: Crystallographic and NMR Studies on a (η1-3,3-Diphenylallyl)-(phosphinooxazoline)palladium Complex".
Organometallics 2002, 21, 4771-4775. |
|
| |
| 114. |
B. Bartels, C. Garcia-Yebra, F. Rominger, G. Helmchen:
"Iridium-Catalysed Allylic Substitution: Stereochemical Aspects and Isolation of IrIII Complexes Related to the Catalytic Cycle".
Eur. J. Inorg. Chem. 2002, 2569-2586.
|
| |
| 113. |
M. Kollmar, H. Steinhagen, J. Janssen, B. Goldfuss, S. Malinovskaya, J. Vazquez, F. Rominger, G. Helmchen:
"(η3-Phenylallyl) (phosphanyloxazoline)palladium Complexes: X-Ray Crystallographic Studies, NMR Investigastions, and Ab Initio/DFT Calculations".
Chem. Eur. J. 2002, 8(14), 3103-3114.
|
| |
| 112. |
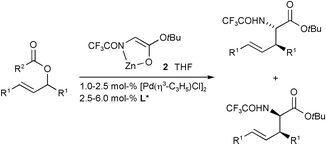
T. Weiß, G. Helmchen, U. Kazmaier:
"Synthesis of amino acid derivatives via enantio- and diastereoselective Pd-catalyzed allylic substitutions with non-stabalized enolate as nucleophile".
Chem. Comm. 2002, 1270-1271. |
|
| |
| 111. |
M. Ostermeier, J. Prieß, G. Helmchen:
"Mono- and Bidentate Phosphinanes - New Chiral Ligands and Their Application in Catalytic Asymmetric Hydrogenations".
Angew. Chem. Int. Ed. 2002, 41, 612-614.
"Ein- und zweizähnige Phosphinane – neue chirale Liganden und ihr Einsatz in der katalytischen asymmetrischen Hydrierung".
Angew. Chem. 2002, 114, 625-628.
|
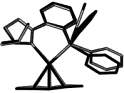
| |
| 110. |
J. Vazquez, B. Goldfuss, G. Helmchen:
"Isomerism of (π-1,3-dimethylallyl)(phosphinooxazoline)Pd complexes: a comparison between experiment and theory".
J. Organomet. Chem. 2002, 641, 67–70. |
|
| |
| 109. |
M. Kollmar, R. Parlitz, S. Oevers, G. Helmchen:
"2-Amino-fluorobenzoic Acid".
Organic Syntheses 2002, 79, 196–203.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 108. |
M. Kollmar, B. Goldfuss, M. Reggelin, F. Rominger, G. Helmchen:
"(Phosphanyloxazoline)palladium Complexes, Part I: (η3-1,3-Dialkylallyl) (phosphanyloxazoline)palladium Complexes: X-Ray Crystallographic Studies, NMR Investigations, and Quantum-Chemical Calculations".
Chem. Eur. J. 2001, 7(22), 4913-4927.
|
| |
| 107. |
H. T. Rong, S. Frey, Y.-J. Yang, M. Zharnikov, M. Buck, M. Wühn, Ch. Wöll, G. Helmchen:
"On the Importance of the Head Group Substrate Bond in Thiol Monolayers: A Study of Biphenyl Based Thiols on Gold and Silver".
Langmuir 2001, 17, 1582-1593.
|
| |
| 106. |
R. Stürmer, B. Schäfer, V. Wolfart, H. Stahr, U. Kazmaier, G. Helmchen:
"A Short and Efficient Synthesis of (S)-1-Boc-2,5-dihydro-1H-pyrrole-2-carboxylic Acid".
Synthesis 2001, 46-48.
|
| |
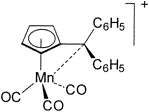
| 105. |
M. A. O. Volland, S. Kudis, G. Helmchen, I. Hyla-Kryspin, F. Rominger, R. Gleiter:
"Structure and Bonding Properties of the Complex (η5-Diphenylfulvene) Mn(CO)3+".
Organometallics 2001, 20, 227-230. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
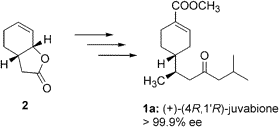
| 104. |
E. J. Bergner, G. Helmchen:
"Enantioselective Synthesis of (+)-Juvabione".
J. Org. Chem. 2000, 65, 5072-5074. |
|
| |
| 103. |
G. Helmchen, A. Pfaltz:
"Phosphinooxazolines - A New Class of Versitale, Modular P,N-Ligands for Asymmetric Catalysis".
Acc. Chem. Res. 2000, 33, 336-345.
|
| |
| 102. |
E. Bergner, G. Helmchen:
"Synthesis of Enantiomerically Pure (-)-Wine Lactone Based on a Palladium-Catalyzed Enantioselective Allylic Substitution".
Eur. J. Org. Chem. 2000, 419-423.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 101. |
S. Schleich, G. Helmchen:
"Pd-Catalyzed Asymmetric Allylic Alkylation of 3-Acetoxy-N-(tert-butyloxycarbonyl)-1,2,3,6-tetrahydropyridine - Preparation of Key Intermediates for Natural Product Synthesis".
Eur. J. Org. Chem. 1999, 2515-2521.
|
| |
| 100. |
G. Helmchen, H. Steinhagen, S. Kudis:
"Enantioselective Catalysis of Allylic Substitutions with Palladium Complexes of Phosphinooxazolines"
in (S. Murahashi, S. G. Davies, Ed.), "Transition Metal Catalysed Reactions - Chemistry for the 21st Century Monographs", Blackwell, Oxford, 1999, pp 241-260. |
| |
| |
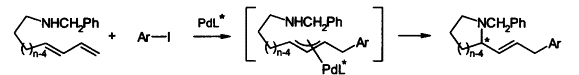
| 99. |
D. Flubacher, G. Helmchen:
"Enantioselective Domino Heck-Allylic Amination Reactions".
Tetrahedron Lett. 1999, 40, 3867-3868. |
|
| |
| 98. |
G. Helmchen:
"Enantioselective palladium catalysed allylic substitutions with asymmetric chiral ligands".
J. Organomet. Chem. 1999, 576, 203-215. |
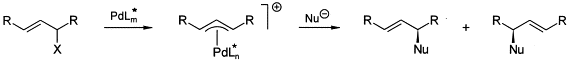
|
| |
| 97. |
B. Bartels, G. Helmchen:
"Ir-catalysed allylic substitution: mechanistic aspects and asymmetric synthesis with phosphorus amidites as ligands".
J. Chem. Soc., Chem. Commun. 1999, 741-742.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 96. |
S. Kudis, H. Steinhagen, G. Helmchen:
"Enantioselective Catalysis of Allylic Substitutions - Fundamentals and Applications".
Chem. Listy 1998, 92, 249-250.
|
| |
| 95. |
S. Kudis, G. Helmchen:
"Enantioselective Allylic Substitution of Cyclic Substrates by Catalysis with Palladium Complexes of P,N-Chelate Ligands with a Cymantrene Unit".
Angew. Chem. Int. Ed. 1998, 37, 3047-3050.
"Enantioselektive allylische Substitution an cyclischen Substraten unter Katalyse mit Palladiumkomplexen von P,N-Chelatliganden mit Cymantreneinheit".
Angew. Chem.. 1998, 110, 3210-3212.
|
| |
| 94. |
M. Johannsen, K. A. Jørgensen, G. Helmchen:
"Synthesis and Application of the first Chiral and Highly Lewis Acidic Silyl Cationic Catalyst".
J. Am. Chem. Soc. 1998, 120, 7637-7638.
|
| |
| 93. |
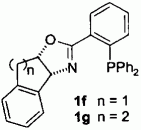
B. Wiese, G. Helmchen:
"Chiral Phosphinooxazolines with a Bi- or Tricyclic Oxazoline Moiety - Applications in Pd-Catalyzed Allylic Alkylations".
Tetrahedron Lett. 1998, 5727-5730. |
|
| |
| 92. |
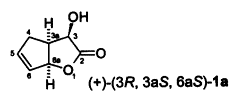
S. Kudis, G. Helmchen:
"First Enantioselective Synthesis of (+)(3R,3aS,6aS)-3-Hydroxy- 3,3a,4,6a-tetrahydrocyclopenta[b]furan-2-one - a Versatile Chiral Heterocyclic Building Block".
Tetrahedron 1998, 54, 10449-10456. |
|
| |
| 91. |
G. Helmchen, H. Steinhagen, M. Reggelin, S. Kudis:
"Asymmetric Allylic Substitutions with Pd Complexes of Phosphinooxazolines as Ligands - Preparative and Mechanistic Aspects".
in Selective Reactions of Metal-Activated Molecules, H. Werner, P. Schreier (Hrsg.), Vieweg, 1998, S. 105-115. |
| |
| |
| 90. |
Ingo Sagasser, Günter Helmchen:
"(Phosphino-oxazoline)copper(II) Complexes as Chiral Catalysts for Enantioselective Diels-Alder Reactions".
Tetrahedron Lett. 1998, 261-264.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 89. |
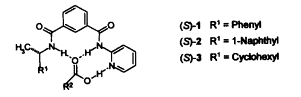
A. Bilz, Th. Stork, G. Helmchen:
"New Chiral Solvating Agents for Carboxylic Acids: Discrimination of Enantiotopic Nuclei and Binding Properties".
Tetrahedron: Asymmetry 1997, 8, 3999-4002. |
|
| |
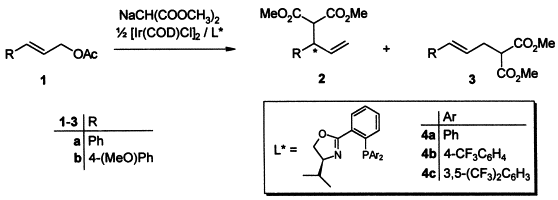
| 88. |
J.P. Janssen, G. Helmchen:
"First Enantioselective Alkylations of Monosubstituted Allylic Acetates Catalyzed by Chiral Iridium Complexes".
Tetrahedron Lett. 1997, 8025-8026. |
|
| |
| 87. |
H. Steinhagen, M. Reggelin, G. Helmchen:
"Palladium-Catalyzed Allylic Alkylation with Phosphinoaryldihydrooxazole Ligands: First Evidence and NMR Spectroscopic Structure Determination of a Primary Olefin-Pd0 Complex".
Angew. Chem. Int. Ed. 1997, 36, 2108-2110.
"Pd-Komplex-katalysierte allylische Alkylierung mit Phosphanylaryldihydrooxazol-Liganden: Erster Nachweis und NMR-spektroskopische Strukturbestimmung eines primären Olefin-Pd0-Komplexes".
Angew. Chem.. 1997, 109, 2199-2202. |
|
| |
| 86. |
G. Helmchen, B. Wiese:
"Efficient Synthesis of New Phosphinooxazoline Ligands from Enantiomerically Pure Amino Alcohols and Diols".
Chimia 1997, 468.
|
| |
| 85. |
G. Helmchen:
"Glossary of Problematic Terms in Organic Stereochemistry".
Enantiomer 1997, 2, 315-318.
|
| |
| 84. |
G. Helmchen, S. Kudis, P. Sennhenn, H. Steinhagen:
"Enantioselective Catalysis with Complexes of Asymmetric P,N-Chelate Ligands".
Pure Appl. Chem. 1997, 69, 513-518.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 83. |
Th. Langer, M. Illich, G. Helmchen:
"Diastereoselective Oxidative Coupling of Enolates of 3-Phenylpropionic Acid Derivatives - EPC Synthesis of ent-Hinokinin".
Synlett 1996, 1137-1139.
|
| |
| 82. |
H. Steinhagen, G. Helmchen:
"Asymmetric Two-Center Catalysis - Learning from Nature".
Angew. Chem. Int. Ed. 1996, 35, 2339-2342.
"Asymmetrische Zweizentren-Katalyse – von der Natur lernen".
Angew. Chem. 1996, 108, 2489-2492.
|
| |
| 81. |
Th. Langer, J. Janssen, G. Helmchen:
"Catalytic Asymmetric Hydrosilylation of Ketones Using Rhodium-(I)-Complexes of Chiral Phosphinooxazoline Ligands".
Tetrahedron, Asymmetry 1996, 1599-1602. |
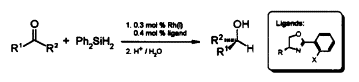
|
| |
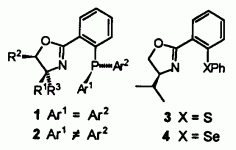
| 80. |
M. Peer, J.C. deJong, M. Kiefer, Th. Langer, H. Rieck, H. Schell, P. Sennhenn, J. Sprinz, H. Steinhagen, B. Wiese, G. Helmchen:
"Preparation of Chiral Phosphorus, Sulfur and Selenium Containing 2-Aryloxazolines".
Tetrahedron 1996, 52, 7547-7583. |
|
| |
| 79. |
Th. Langer, G. Helmchen:
"Highly Efficient New Catalysts for Enantioselective Transfer Hydrogenation of Ketones".
Tetrahedron Lett. 1996, 1381-1384. |
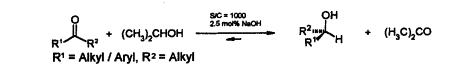
|
| |
| 78. |
E. Urban, G. Knühl, G. Helmchen:
"Enantiomerically Pure 5-Substituted 2-Oxo-cyclopentanecarboxylates by Conjugate Addition of Cuprates to Asymmetric Shielded 2-Oxo-cyclopentenecarboxylates".
Tetrahedron 1996, 52, 971-986.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
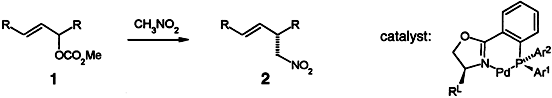
| 77. |
H. Rieck, G. Helmchen:
"Palladium Complex Catalyzed Asymmetric Allylic Substitutions with Nitromethane: Enantioselectivities Exceeding 99 %ee".
Angew. Chem. Int. Ed. 1995, 34, 2687-2689.
"Palladiumkomplex-katalysierte asymmetrische allylische Substitutionen mit Nitromethan: Enantioselektivität mit ee-Werten von über 99.9%".
Angew. Chem.1995, 107, 2881-2883. |
|
| |
| 76. |
E. Urban, G. Knühl, G. Helmchen:
"A Short Synthesis of (-)-Chokol A".
Tetrahedron 1995, 51, 13031-13038.
|
| |
| 75. |
E. Urban, G. Knühl, G. Helmchen:
"EPC Synthesis of 5-Substituted 2-Oxo-cyclopentanecarboxylates via Conjugate Addition of Cuprates to Asymmetrically Shielded 2-Oxo-cyclopentanecarboxylates".
Tetrahedron Lett. 1995, 7229-7232.
|
| |
| 74. |
G. Knühl, P. Sennhenn, G. Helmchen:
"New Chiral β-Phosphinocarboxylic Acids and their Application in Palladium-catalysed Asymmetric Allylic Alkylations".
J. Chem. Soc., Chem. Commun. 1995, 1845-1846.
|
| |
| 73. |
Th. Langer, M. Illich, G. Helmchen:
"Diastereoselective Oxidative Coupling of Enolates of Chiral Carboxylic Acid Derivatives".
Tetrahedron Lett. 1995, 4409-4412. |
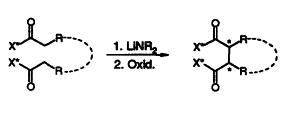
|
| |
| 72. |
Th. Stork, G. Helmchen:
"A New Chiral Solvating Agent for Carboxylic Acids Based on Directed Hydrogen Bonding".
Recl. Trav. Chim. Pays-Bas, 1995, 114, 253-254.
|
| |
| 71. |
G. Helmchen, J. Janssen, M. Kiefer, M. Peer, H. Rieck, P. Sennhenn, J. Sprinz:
"Enantioselective Catalysis of Allylic Substitutions with Pd Complexes of Phosphinooxazolines".
Electronic Conference on Trends in Organic Chemistry (ECTOC-1), June 12-July 7, 1995
Eds. H. S. Rzepa, C. Leach, J. M. Goodman (CD-ROM), The Royal Society of Chemistry, 1996; ISBN 0 85404 899 5.
|
| |
| 70. |
G. Helmchen:
"Vocabulary and Nomenclature of Organic Stereochemistry".
in Houben-Weyl E21a, Stereoselective Synthesis
G. Helmchen, R.W. Hoffmann, J. Mulzer, E. Schaumann (Hrsg.), 1995, 1-74. |
| |
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 69. |
E. Urban, G. Knühl, G. Helmchen:
"Eine effiziente Synthese von ()-Chokol A".
Sci. Pharm. 1994, 62, 195.
|
| |
| 68. |
P. Sennhenn, B. Gabler, G. Helmchen:
"Enantiomerically Pure Cycloalkenylacetic Acid Derivatives via Pd-Catalyzed Asymmetric Allylic Alkylation and Subsequent Enantiomeric Enrichment via Iodolactones".
Tetrahedron Lett. 1994, 8595-8598. |
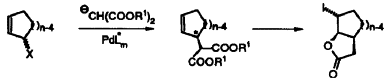
|
| |
| 67. |
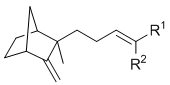
A. Krotz, G. Helmchen:
"Total Syntheses, Optical Rotations and Fragrance Properties of Sandalwood Fragrances, (-)-(Z)- and (-)-(E)-β-Santalol and their Enantiomers, ent-β-Santalene".
Liebigs Ann. Chem. 1994, 601-609. |
|
| |
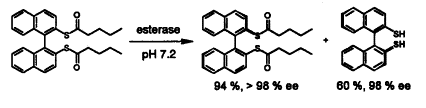
| 66. |
M. Kiefer, R. Vogel, G. Helmchen, B. Nuber:
"Resolution of (1,1´-Binaphthalene)-2,2´-dithiol by Enzyme Catalysed Hydrolysis of a Racemic Diacyl Derivative".
Tetrahedron 1994, 50, 7109-7114. |
|
| |
| 65. |
P. von Matt, O. Loiseleur, G. Koch, A. Pfaltz, C. Lefeber, T. Feucht, G. Helmchen:
"Enantioselective Allylic Amination with Chiral (Phosphino-oxazoline)Pd Catalysts".
Tetrahedron: Asymmetry 1994, 5, 573-584. |
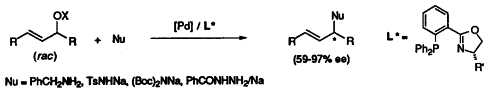
|
| |
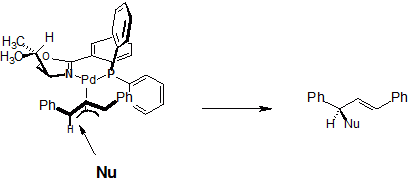
| 64. |
J. Sprinz, M. Kiefer, G. Helmchen, M. Reggelin, G. Huttner, O. Walter, L. Zsolnai:
"Catalysis of Allylic Substitutions by Pd Complexes of Oxazolines Containing an Additional P, S or Se Center. X-Ray Crystal Structures and Solution Structures of Chiral ω-Allyl Complexes of Phosphinoaryloxazolines".
Tetrahedron Lett. 1994, 1523-1526. |
|
| |
| 63. |
H. Heidel, G. Huttner, R. Vogel, G. Helmchen:
"Ein neuartiger chiraler Synthesebaustein mit Neopentangerüst, Chemo-enzymatische Darstellung von (R)-CH3C(CH2OSO2CF3)(CH 2Cl)(CH2Br)".
Chem. Ber. 1994, 127, 271-274.
|
| |
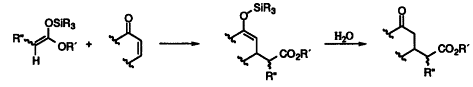
| 62. |
V. Berl, G. Helmchen, S. Preston:
"Highly Effective Catalysts for the Conjugate Addition of Silyl Ketene Acetals to Enones (Mukaiyama-Michael Reaction)".
Tetrahedron Lett. 1994, 233-236. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 61. |
C.E. Dempfle, K. Hochreuter, G. Helmchen, A. Leinweber, D.L. Heene:
"Microtiter assay for factor VIII activity".
in (Hrsg.: J. McDonagh, R. Seitz, R. Egbring) Factor XIII, Schattauer, Stuttgart, 1993, pp 112-121. |
| |
| |
| 60. |
H. Heidel, G. Huttner, G. Helmchen:
"Tripodliganden mit drei verschiedenen Donorgruppen: Synthese und Koordination von CH3C(CH2PR2)(CH2PR´2 )(CH2PR´´2)".
Z. Naturforsch. B 1993, 48, 1681-1692.
|
| |
| 59. |
G. Helmchen, A. Krotz, H.-P. Neumann, M. L. Ziegler:
"Enantiomerically Pure Chiral Building Blocks for Synthesis of Carbocyclic Nucleoside Analogues, A Formal Synthesis of Aristeromycin".
Liebigs Ann. Chem. 1993, 1313-1317.
|
| |
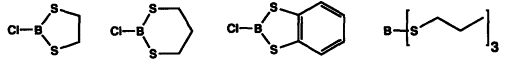
| 58. |
J. Howarth, G. Helmchen, M. Kiefer:
"Lewis Acids Based on Sulfur Containing Boron Heterocycles and Trialkylthioboranes".
Tetrahedron Lett. 1993, 34, 4095-4096. |
|
| |
| 57. |
P. Kramp, G. Helmchen, A.B. Holmes:
"Syntheses of ent-Multifidene and Related Compounds".
J. Chem. Soc., Chem. Commun. 1993, 551-552.
|
| |
| 56. |
J. Sprinz, G. Helmchen:
"Phosphinoaryl- and Phosphinoalkyloxazolines as New Chiral Ligands for Enantioselective Catalysis: Very High Enantioselectivity in Palladium Catalyzed Allylic Substitutions".
Tetrahedron Lett. 1993, 1769-1772 |
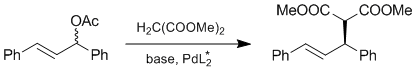
|
| |
| 55. |
K.-L. Gibis, G. Helmchen, G. Huttner, L. Zsolnai:
"Enantiomerically Pure C2-Symmetric Bridged Ferrocene and Titanocene Derivatives".
J. Organomet. Chem. 1993,445, 181-186.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 54. |
P. Hamley, G. Helmchen, A. B. Holmes, D. R. Marshall, J. W. M. MacKinnon, D. F. Smith, J. W. Ziller:
"Diastereoselective Imino Ester Cycloadditions. Enantioselective Synthesis of Azabicyclo[2.2.1]heptenes".
J. Chem. Soc., Chem. Commun. 1992, 786-788.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 53. |
D. Sartor, J. Saffrich, C. Richards, H. Lambert, G. Helmchen:
"Enantioselective Diels-Alder Reactions of Enals: Fighting Species Multiplicity of the Catalyst with Donor Solvents".
Tetrahedron: Asymmetry 1991, 2, 639-642.
|
| |
| 52. |
G. Helmchen, A. Goeke, S. Kreisz, A. Krotz, G. Lauer, G. Linz:
"Cyclopentanoid Natural Products via Asymmetric Diels-Alder Reactions"
in Atta-ur-Rahman, Studies in Natural Product Chemistry, Vol. 8, Elsevier, Amsterdam 1991, 139-158. |
| |
| |
| 51. |
G. Helmchen, A. Krotz, K.-T. Ganz, D. Hansen:
"C2-Symmetric Bioxazolines and Bithiazoline as New Chiral Ligands for Metal Ion Catalyzed Asymmetric Syntheses: Asymmetric Hydrosilylation".
Synlett 1991, 257-259.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 50. |
A. Krotz, G. Helmchen:
"Total Syntheses of Sandalwood Fragrances: (Z)- and (E)-β-Santalol and their Enantiomers, ent-β-Santalene".
Tetrahedron: Asymmetry 1990, 537-540.
|
| |
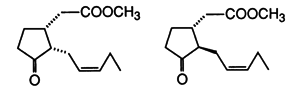
| 49. |
G. Helmchen, A. Goeke, G. Lauer, M. Urmann, J. Fries:
"Building Blocks for the Synthesis of Enantiomerically Pure Jasmonoids: Synthesis of (+)-Methyl Epijasmonate".
Angew. Chem. Int. Ed. 1990, 29, 1024-1025.
"Bausteine zur Synthese von enantiomerenreinen Jasmonoiden: Synthese von (+)-Methylepijasmonat".
Angew. Chem. 1990, 102, 1079-1081. |
|
| |
| 48. |
D. Sartor, J. Saffrich, G. Helmchen:
"Enantioselective Diels-Alder Additions with New Chiral Lewis Acids Derived from Amino Acids".
Synlett 1990, 197-198.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
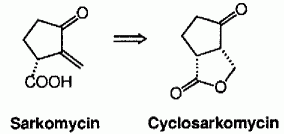
| 47. |
G. Helmchen, G. Linz, J. Weetman, A. F. Abdel Hady:
"Asymmetric Diels-Alder Reactions: EPC-Synthesis of a Stable Sarkomycin Precursor (Cyclosarkomycin)".
Tetrahedron Lett. 1989, 5599-5602. |
|
| |
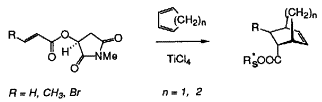
| 46. |
T. Poll, A. F. Abdel Hady, R. Karge, J. Weetman, G. Helmchen:
"N-Substituted Hydroxysuccinimides from (S)-Malic Acid as New Reagents for Asymmetric Diels-Alder Additions to Enoates".
Tetrahedron Lett. 1989, 5595-5598. |
|
| |
| 45. |
G. Helmchen, A. F. Abdel Hady, H. Hartmann, R. Karge, A. Krotz, K. Sartor, M. Urmann:
"Natural Product Syntheses Based on Asymmetric Diels-Alder Reactions".
Pure Appl. Chem. 1989, 61, 409-412.
|
| |
| 44. |
M. Toth, G. Helmchen, U. Leikauf, Gy. Sziraki, G. Szöcs:
"Behavioral Activity of Stereoisomers of 5,9-Dimethyl-heptadecanes, the Sex Pheromone of Leucoptera scitella L. (Lepidoptera: Lyonetidae)".
J. Chem. Ecol. 1989, 15, 1535-1543.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
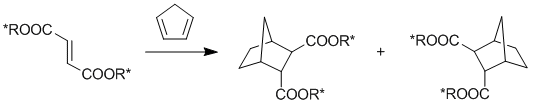
| 43. |
H. Hartmann, A. F. Abdel Hady, K. Sartor, J. Weetman, G. Helmchen:
"High Stereoselectivity of Lewis-Acid-Catalyzed and Uncatalyzed Diels-Alder-Reactions of the Fumarate of (S)-Ethyl Lactate".
Angew. Chem. Int. Ed. 1987, 26, 1143-1145.
"Hohe Stereoselektivität bei Lewis-Säure-katalysierten und bei unkatalysierten Diels-Alder-Reaktionen des Fumarsäureesters von (S)-Ethyllactat".
Angew. Chem. 1987, 99, 1188-1189. |
|
| |
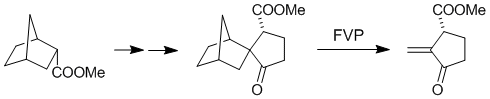
| 42. |
G. Helmchen, K. Ihrig, H. Schindler:
"EPC-Syntheses via Asymmetric Diels-Alder Reactions/Retro Diels-Alder Reactions I: (R)- and (S)-Matsutake Alkohol, (R)- and (S)-Sarkomycin Methyl Ester".
Tetrahedron Lett. 1987, 183-186. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 41. |
G. Helmchen, R. Karge, J. Weetman:
"Asymmetric Diels-Alder Reactions with Chiral Enoates as Dienophiles".
in R. Scheffold (Ed.), Modern Synthetic Methods 1986, Vol. 4, p.262-306, Springer, Heidelberg 1986. |
| |
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 40. |
G. Helmchen, G. Wegner:
"Asymmetric Synthesis of β-Substituted Alcanoic Acids via Highly Stereoselective Conjugate Additions of Organocopper Compounds to Chiral Enoates".
Tetrahedron Lett. 1985, 6051-6054.
|
| |
| 39. |
G. Helmchen, G. Wegner:
"Synthesis of Enantiomerically Pure (S)-3-Trichloromethylbutyric Acid Via Asymmetric Conjugate Addition of Trichloromethyl Metal Compounds to a Chiral Enoate. Activation Effect of a Sulfonylamino Group".
Tetrahedron Lett. 1985, 6047-6050.
|
| |
| 38. |
G. Helmchen, U. Leikauf, I. Taufer-Knöpfel:
"Enantio- and anti-Diastereoselective Aldol Additions of Acetates and Propionates via O-Silyl Ketene Acetales".
Angew. Chem. Int. Ed. 1985, 24, 874-875.
"Enantio- und anti-diastereoselektive Aldoladditionen von Acetaten und Propionaten über O-Silylketenacetale".
Angew. Chem. 1985, 97, 874-876.
|
| |
| 37. |
G. Helmchen:
"Stereoselection at 3-Carbon Units. Recent Studies and Applications in Natural Product Synthesis"
in J. Streith, H. Prinzbach, G. Schill (Eds.), Organic Synthesis, an Interdisciplinary Challenge, p. 167-177, Blackwell, Oxford 1985. |
| |
| |
| 36. |
D. Dorsch, E. Kunz, G. Helmchen:
"Syntheses of Dictyopterene B (Hormosirene) and its Enantiomer via Asymmetric ScN' Reactions".
Tetrahedron Lett. 1985, 3319-3322.
|
| |
| 35. |
T. Poll, A. Sobczak, H. Hartmann, G. Helmchen:
"Diastereoface-discriminative Metal Coordination in Asymmetric Synthesis: D-Pantolactone as Practical Chiral Auxiliary for Lewis Acid Catalyzed Diels-Alder Reactions".
Tetrahedron Lett. 1985, 3095-3098.
|
| |
| 34. |
T. Poll, J. O. Metter, G. Helmchen:
"Concerning the Mechanism of the Asymmetric Diels-Alder Reaction: First Crystal Structure Analysis of a Lewis Acid Complex of a Chiral Dienophile".
Angew. Chem. Int. Ed. 1985, 24, 112-114.
"Zum Mechanismus der asymmetrischen Diels-Alder-Reaktion: Erste Kristallstrukturanalyse eines Lewis-Säure-Komplexes eines chiralen Dienophils".
Angew. Chem. 1985, 97, 116-118.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
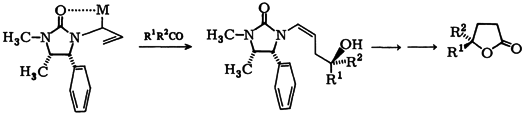
| 33. |
H. Roder, G. Helmchen, E.-M. Peters, K. Peters, H.-G. von Schnering:
"Highly Enantioselective Homoaldol Additions with Chiral N-Allylureas - Application to the Synthesis of Optically Pure γ-Lactones".
Angew. Chem. Int. Ed. 1984, 23, 898-899.
"Hochenantioselektive Homoaldol-Addition mit chiralen N-Allylharnstoffen - Anwendung zur Synthese optisch reiner γ-Lactone".
Angew. Chem. 1984, 96, 895-896. |
|
| |
| 32. |
T. Poll, G. Helmchen, B. Bauer:
"Influence of Polar Groups in Thermal and Lewis Acid Promoted Asymmetric Diels-Alder Additions: Lactic Acid Derivatives as Practical Highly Selective and Configurationally Dichotomic Reagents".
Tetrahedron Lett. 1984, 2191-2194.
|
| |
| 31. |
G. Helmchen, R. Wierzchowski:
"Preparation of Enantiomerically Pure Chiral Alcohols by Asymmetric Alkylation of Glycolates".
Angew. Chem. Int. Ed. 1984, 23, 60-61.
"Herstellung enantiomerenreiner chiraler Alkohole durch asymmetrische Alkylierung von Glykolaten".
Angew. Chem. 1984, 96, 59-60.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 30. |
G. Helmchen, A. Selim, D. Dorsch, I. Taufer:
"Influence of Cation Complexing Solvent Additives and Functional Groups on Asymmetric Alkylations of Esters via Lithium Enolates".
Tetrahedron Lett. 1983, 3213-3216.
|
| |
| 29. |
G. Helmchen, R. Schmierer:
"A Total Synthesis of Enantiomerically Pure Vitamin E Side Chain Using a Chiral Propionate Synthon".
Tetrahedron Lett. 1983, 1235-1238.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 28. |
V. Prelog, G. Helmchen:
"Basic Principles of the CIP-System and Proposals for a Revision".
Angew. Chem. Int. Ed. 1982, 21, 567-583.
"Grundlagen des CIP-Systems und Vorschläge für eine Revision".
Angew. Chem. 1982, 94, 614-631.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 27. |
G. Helmchen:
"Asymmetrische Synthesen mit bioanalogen Synthons"
Jahrbuch der Akademie der Wissenschaften in Göttingen 1981, S. 19-27.
|
| |
| 26. |
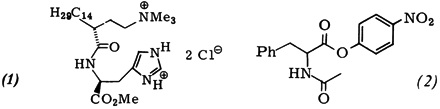
J. M. Brown, R. L. Elliott, C. G. Griggs, G. Helmchen, G. Nill:
"Selective Micellar Catalysis with Histidinyl Surfactants of Defined Absolute Configuration".
Angew. Chem. Int. Ed. 1981, 20, 890-892.
"Selektive micellare Katalyse mit Histidinyl-Tensiden definierter absoluter Konfiguration".
Angew. Chem. 1981, 93, 906-907. |
|
| |
| 25. |
G.Helmchen, P. A. Langley:
"Bekämpfung der Tsetse-Fliege mit Pheromonen".
Nachr. Chem. Tech. Lab. 1981, 29, 294-299.
|
| |
| 24. |
R. Schmierer, G. Grotemeier, G. Helmchen, A. Selim:
"Functional Groups at Concave Sites: Asymmetric Alkylation of Esters with Very High Stereoselectivity and Reversal of Configuration by Change of Solvent".
Angew. Chem. Int. Ed. 1981, 20, 207-208.
"Funktionelle Gruppen in konkaver Lage: Asymmetrische Alkylierung von Estern mit sehr hoher Stereoselektivität und Konfigurationsumkehrung durch Solvensänderung".
Angew. Chem. 1981, 93, 209-211.
|
| |
| 23. |
G. Helmchen, R. Schmierer:
"Functional Groups at Concave Sites: Asymmetric Diels-Alder Synthesis with Almost Complete (Lewis-Acid Catalyzed) or High (Uncatalyzed) Stereoselectivity".
Angew. Chem. Int. Ed. 1981, 20, 205-207.
"Funktionelle Gruppen in konkaver Lage: Asymmetrische Diels-Alder-Synthese mit nahezu vollständiger (Lewis-Säure-katalysiert) und hoher (unkatalysiert) Stereoselektivität".
Angew. Chem. 1981, 93, 208-209.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 22. |
E. Ade, G. Helmchen, G. Heiligenmann:
"Syntheses of the Stereoisomers of 17,21-Dimethylheptatriacontane - Sex Recognition Pheromone of the Tsetse Fly".
Tetrahedron Lett. 1980, 1137-1140.
|
| |
| 21. |
G. Helmchen:
"Kernresonanz-Diastereotopieeffekte und flüssigkeitschromatographische Trennung von diastereomeren Carbonsäurederivaten - Eine Untersuchung der methodischen und theoretischen Grundlagen am Beispiel von Carbonsäureamiden.".
Habilitationsschrift zur Erlangung der Lehrberechtigung (venia legendi) für Organische Chemie an der Universität Stuttgart, 1980.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 20. |
B. Glatz, G. Helmchen, H. Muxfeldt, H. Porcher, R. Prewo, J. Senn, J. J. Stezowski, R. J. Stojda, D. R. White:
"A Total Synthesis and Structural Aspects of Racemic 8-Oxygenated Tetracyclines".
J. Am. Chem. Soc. 1979, 101, 2171-2181.
|
| |
| 19. |
G. Helmchen, G. Nill:
"Preparative Scale Directed Resolution of Enantiomeric Amines via Liquid Chromatography of Diastereomeric 4-Hydroxybutyramides".
Angew. Chem. Int. Ed. 1979, 18, 65-66.
"Gezielte präparative Trennung enantiomerer Amine via Flüssigkeitschromatographie diastereomerer 4-Hydroxybutyramide".
Angew. Chem. 1979, 91, 66-68.
|
| |
| 18. |
G. Helmchen, G. Nill, D. Flockerzi, M. S. K. Youssef:
"Preparative Scale Directed Resolution of Enantiomeric Carboxylic Acids and Lactones via Liquid Chromatography and Neighboring-Group Assisted Hydrolysis of Diastereomeric Amides".
Angew. Chem. Int. Ed. 1979, 18, 63-65.
"Gezielte präparative Trennung enantiomerer Carbonsäuren und Lactone via Flüssigkeitschromatographie und nachbargruppenunterstützte Hydrolyse diastereomerer Amide".
Angew. Chem. 1979, 91, 65-66.
|
| |
| 17. |
G. Helmchen, G. Nill, D. Flockerzi, W. Schühle, M. S. K. Youssef:
"Extreme Liquid Chromatographic Separation Effects in the Case of Diastereomeric Amides Containing Polar Substituents".
Angew. Chem. Int. Ed. 1979, 18, 62-63.
"Extreme flüssigkeitschromatographische Trenneffekte bei diastereomeren Amiden mit polaren Substituenten".
Angew. Chem. 1979, 91, 64-65.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 16. |
B. Glatz, G. Helmchen:
"Ein apparativ einfaches System und Säulen höchster Trennleistung zur präparativen Mitteldruck-Flüssigkeitschromatographie".
Universität Stuttgart 1978, Anhang I der Habilitationsarbeit von Prof. Dr. G. Helmchen.
|
| |
| 15. |
H. Völter, G. Helmchen:
"Enantiomers as a Result of Restricted Rotation about Partial Double Bonds: Resolution of N-Nitroso-, (N-Formyl-), and N-Thioformyl-4-piperidinecarboxylic Acids".
Tetrahedron Lett. 1978, 1251-1254.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 14. |
G. Helmchen:
"Präparative Trennung von Stereoisomeren - Gezielte Enantiomerentrennung über diastereomere Amide"
2. Deutsche Diskussionstagung für Anwender der Hochdruck-Flüssigkeitschromatographie, 26.-28. Oktober 1977, Königstein/ Taunus, Handbuch S. 85-99.
|
| |
| 13. |
H. Gölz, B. Glatz, G. Haas, G. Helmchen, H. Muxfeldt:
"N-Acyl-N'-arylsulfonyldiazenes; Detektion and Use in the Synthesis of Amides".
Angew. Chem. Int. Ed. 1977, 16, 728-729.
"N-Acyl-N'-arylsulfonyldiazene; Nachweis und Verwendung zur Synthese von Amiden".
Angew. Chem. 1977, 89, 742-743.
|
| |
| 12. |
G. Helmchen, H. Völter, W. Schühle:
"Directed Resolution of Enantiomers via Liquid Chromatography of Diastereomeric Derivatives III. A Convenient Method to Determine the Absolute Configuration of Carboxylic Acids R1R2HCCOOH".
Tetrahedron Lett. 1977, 1417-1420.
|
| |
| 11. |
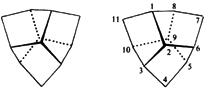
G. Helmchen, G. Staiger:
"Synthesis and Absolute Configuration of Enantiomerically Pure D3-Trishomocubanes (Pentacyclo[6.3.O.O2,6.O3,1O.O5,9]undecane) and Trishomocubanones".
Angew. Chem. Int. Ed. 1977, 16, 116-117.
"Synthese und absolute Konfiguration der enantiomer reinen D3-Trishomocubane (Pentacyclo[6.3.O.O2,6.O3,1O.O5,9]undecan) and Trishomocubanone".
Angew. Chem. 1977, 89, 119-120. |
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 10. |
G. Helmchen, R. Schmierer:
"Determination of the Absolute Configuration of Chiral Thiols by 1H-NMR Spectroscopy of Diastereomeric Thiol Esters".
Angew. Chem. Int. Ed. 1976, 15, 703-704.
"Bestimmung der absoluten Konfiguration von chiralen Thiolen durch 1H-NMR-Spektroskopie diastereomerer Thiolester".
Angew. Chem. 1976, 88, 770-771.
|
| |
| 9. |
H. Muxfeld, W.-D. Unterweger, G. Helmchen:
"Transthioacetalisierung. Eine einfache Methode zur hydrolytischen Spaltung von Thioacetalen".
Synthesis 1976, 694-696.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 8. |
G. Helmchen:
"Über eine neue Methode zur Bestimmung der absoluten Konfiguration von chiralen sekundären Alkoholen und Aminen, NMR-Spektroskopie von diastereomeren Estern und Amiden der α-Phenylbutter- und Hydratropasäure".
Tetrahedron Lett. 1974, 1527-1530.
|
| |
| 7. |
G. Helmchen, W. Strubert:
"Determination of Optical Purity by High Performance Liquid Chromatography".
Chromatographia 1974, 7, 713-715.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 6. |
G. Helmchen, G. Haas, V. Prelog:
"Über die stereoisomeren α-Phenyläthylamide der Biphenyl-2,2',6,6'-tetracarbonsäure, Molekeln der Punktsymmetrie D2, S4, C2 und C1".
Helv. Chim. Acta 1973, 56, 2255-2270.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 5. |
G. Helmchen, R. Ott, K. Sauber:
"Gezielte Trennung und absolute Konfiguration von enantiomeren Carbonsäuren und Aminen".
Tetrahedron Lett. 1972, 3873-3878.
|
| |
| 4. |
G. Helmchen, V.Prelog:
"Über 1,12-Dioxa-[12]paracyclophan-Derivate mit einer Pseudoasymmetrieebene".
Helv. Chim. Acta 1972, 55, 2612-2620.
|
| |
| 3. |
G. Helmchen, V. Prelog:
"Über atropisomere Biphenyl-Derivate mit einer Pseudoasymmetrieachse".
Helv. Chim. Acta 1972, 55, 2599-2611.
|
| |
| 2. |
V. Prelog, G. Helmchen:
"Pseudoasymmetrie in der organischen Chemie".
Helv. Chim. Acta 1972, 55, 2581-2598.
|
| |
| 2025 2023 2021 2020 2019 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 2005 2004 2003 2002 2001 2000 1999 1998 1997 1996 1995 1994 1993 1992 1991 1990 1989 1987 1986 1985 1984 1983 1982 1981 1980 1979 1978 1977 1976 1974 1973 1972 1971 |
| |
| 1. |
G. Helmchen:
"Untersuchungen über 'pseudoasymmetrische' organische Verbindungen".
Abhandlung zur Erlangung der Würde eines Doktors der technischen Wissenschaften der Eidgenössischen Technischen Hochschule Zürich, 1971. |
| |