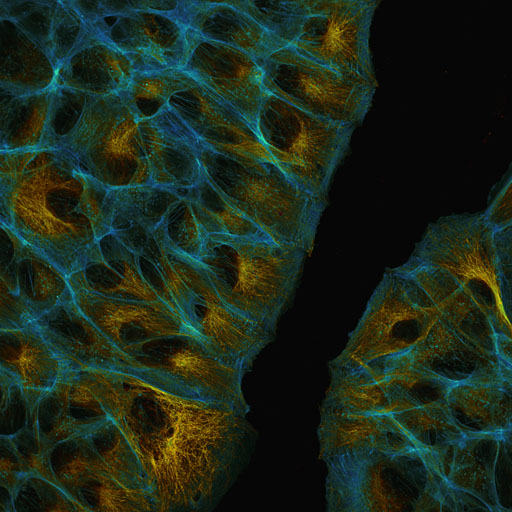
Nikon A1R confocal microscope
The A1R features a so called "hybrid scanner", that is two scanners in one scanning-head: a glavano scanner for low-noise images ranging in resolution from 64 x 64 up to 4096 x 4096 pixels and a resonant scanner that is capable of taking up to 30 full 512 x 512 pixel frames per second. What is more, both scanners can be used in parallel: while the fast resonant scanner is acquiring images the galvano scanner may perform bleaching or activation of user defined regions of interest.
For general imaging, the A1R offers three operating modes. In the standard mode the signal of the fluorochromes that are excited by six available lasers (405, 445, 488, 514, 561 and 640nm) is separated by dichroic mirrors and optical filters. Up to four channels can be acquired in a line mode using two normal and two ultra sensitive GaAsP-detectors. If fluorochromes are used whose emission is too close to be separated by this method (like GFP and YFP), a spectral detector does the job: here the fluorochrome emission can be split up in up to 32 bands of 10 nm bandwidth, detected by a GaAsP-detector. By this these signals can be clearly and reliably distinguished by a process called "spectral unmixing" implemented in the NIS-Elements image acquisition and analysis software. Spectral imaging also makes it possible to separate a fluorescent background from specific signal.
The third mode is called "virtual filters". Here the spectral detector is used to freely define ranges where signals are detected.
The A1R confocal is mounted on a fully automated inverted Nikon Ti2 microscope. A Nikon Perfect Focus System (PFS) is included that continuously determines the distance to the coverslip, readjusting it if required, for instance due to changes because of thermal drift. Long-term experiments are strongly facilitated by this. For live cell imaging there's an on-stage incubation chamber from TokaiHit, controlling temperature, CO2-concentration and humidity.
Objectives
- Nikon Plan Apo λ 4x NA 0.20 (WD 20mm, FOV 3.18 x 3.18mm)
- Nikon Plan Apo λ 10x NA 0.45 (WD 4mm, FOV 1.27 x 1.27mm)
- Nikon Plan Apo λ 20x NA 0.75 (WD 1mm, FOV 0.64 x 0.64mm)
- Nikon Plan Fluor 40x NA 1.3 OI (WD 0.2mm, FOV 0.32 x 0.32mm)
- Nikon N Apo 60x NA 1.4 λs OI (WD 0.14mm, FOV 0.21 x 0.21mm)
- Nikon Plan Apo IR 60x NA 1.27 WI (WD 0.18 - 0.16mm, FOV 0.21 x 0.21mm)
WD = working distance
FOV = field of view @ zoom 1
OI = oil immersion
WI = water immersion
Relative fluorescence image brightness
The higher the NA of an objective, the more light it collects. The higher the magnification on the other hand, the more the collected light is "diluted". The list shows that the 40x objective is the most efficient one in terms of image brightness:
- Nikon Plan Apo λ 4x NA 0.20: 1.0
- Nikon Plan Apo λ 10x NA 0.45: 4.1
- Nikon Plan Apo λ 20x NA 0.7: 7.9
- Nikon Plan Fluor 40x NA 1.3: 17.9
- Nikon Apo λs 60x NA 1.40: 10.7
- Nikon Plan Apo IR 60x NA 1.27: 7.2
Signal Detection
In the "normal" 4 channel detection mode, the signal of the fluorochromes that are excited by the available lasers are detected in the following ranges (center wavelength [nm]/bandwidth [nm]):
- 450/50
- 482/35
- 515/30
- 525/50
- 540/30
- 595/50
- 700/75
In the spectral detection mode signal detection is possible from 400 to 750nm with a spectral discrimination of 10 or 20nm (32 channels maximum).
3D rendering of zebrafish brain acquired with the Nikon A1R confocal (Carlo Beretta).
Dictyostelium amoeba, maximum intensity projection of 5 z-planes in 5s intervals over 3min with A1R resonant scanner. Actin (red) and microtubles (green). Field is 106 x 106 µm (Christian Ackermann).