Welcome to the Straub Group
An octacopper hexaacetylide cluster salt with three bidentate ancillary N-heterocyclic carbene (NHC) ligands was prepared under acidic reaction conditions. This cluster is the first molecular copper acetylide complex that features high activity in the copper-catalyzed azide–alkyne cycloaddition (CuAAC), with added acetic acid even at -5 °C.
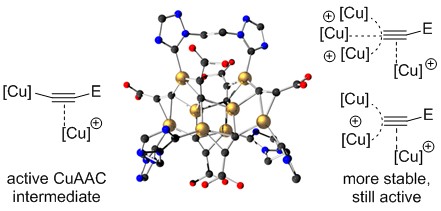
Fluktuierender Kupferacetylid-Cluster in der CuAAC-Katalyse
Fluxional Copper Acetylide Cluster in CuAAC Catalysis
Ata Makarem, Regina Berg, Frank Rominger, Bernd F. Straub*, Angew. Chem. 2015, 127, 7539-7543; Angew. Chem. Int. Ed. 2015, 54, 7431-7435.
The escaping lynx symbolizes the short lifetime of the first detected cationic copper carbene complex without heteroatom stabilization. The snow leopard that breaks a bar of his cage symbolizes the marginal silver-to-carbene back-bonding. Steric shielding by the IPr** ancillary ligand and the carbenes' substituents rendered possible the characterization of the greenish curry-yellow copper carbene complex and of the mint-green silver carbene complex, respectively.


Kupfer- und Silber-Carbenkomplexe ohne Heteroatom-Stabilisierung: Struktur, Spektroskopie und Relativistische Effekte
Copper and Silver Carbene Complexes without Heteroatom-Stabilization: Structure, Spectroscopy, and Relativistic Effects
Matthias W. Hussong, Wilhelm T. Hoffmeister, Frank Rominger, Bernd F. Straub*, Angew. Chem. 2015, 127, 10472-10476; Angew. Chem. Int. Ed. 2015, 54, 10331-10335 (announced as "hot paper", front inside cover).
The captured tiger symbolizes the high electronic reactivity of the first isolated gold carbene complex without heteroatom stabilization. The full characterization of the emerald-green carbene complex reveals a significant Au=C back-bonding. The animated ball-and-stick model structure is derived from a single-crystal X-ray analysis: See gif animation (1024x768 resolution; 11 MB).

Isolierung eines nicht-Heteroatom-stabilisierten Goldcarbens
Isolation of a Non-Heteroatom-Stabilized Gold-Carbene Complex
Matthias W. Hussong, Frank Rominger, Petra Krämer, Bernd F. Straub*, Angew. Chem. 2014, 126, 9526-9529; Angew. Chem. Int. Ed. 2014, 53, 9372-9375 (announced as “hot paper”).
The lipophilic, water-stable, thermostable and cost-efficient aluminates "aletbate" and "aletpate" are halogen-free anions whose salts display a high tendency for crystallization and a high solubility in unpolar organic solvents. They are an environmentally more benign alternative for fluorinated and chlorinated weakly-coordinating anions.
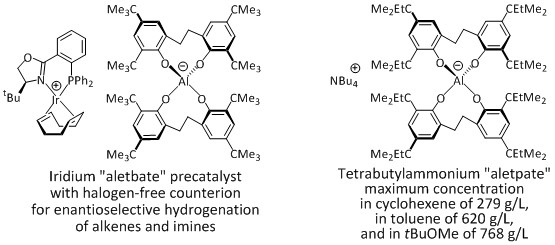
Halogen-free water-stable aluminates as replacement for persistent fluorinated weakly-coordinating anions
Timo Söhner, Felix Braun, Lena Charlotte Over, Sven Mehlhose, Frank Rominger, Bernd F. Straub*, Green Chem. 2014, 16, 4696-4707.
The sterically shielded gold complex IPr**AuNTf2 features remarkably high catalytic activity and stability. Alkyne hydration reactions and phenol syntheses proceed with high to excellent yields.
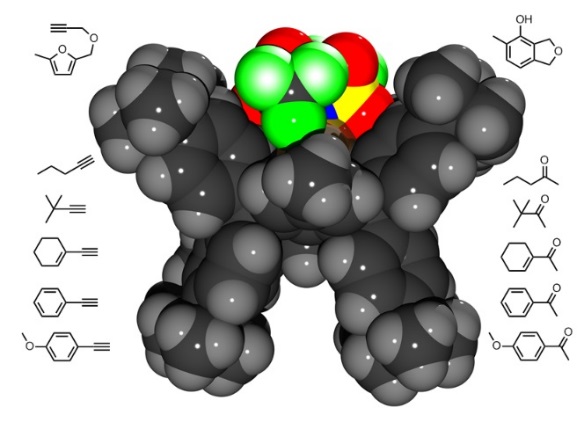
Mechanistic Investigations of a Stable, Highly Active, Extremely Sterically Shielded Molecular Gold Catalyst
Simone G. Weber, David Zahner, Frank Rominger, Bernd F. Straub*, ChemCatChem 2013, 5, 2330-2335.
A dinuclear copper complex features an unprecedented activity in homogeneous CuAAC "click" catalysis. This is the first direct evidence for the presence of two copper(I) atoms in the rate-limiting step in the reaction of organoazides and terminal alkynes to 1,4-disubstituted 1,2,3-triazoles.
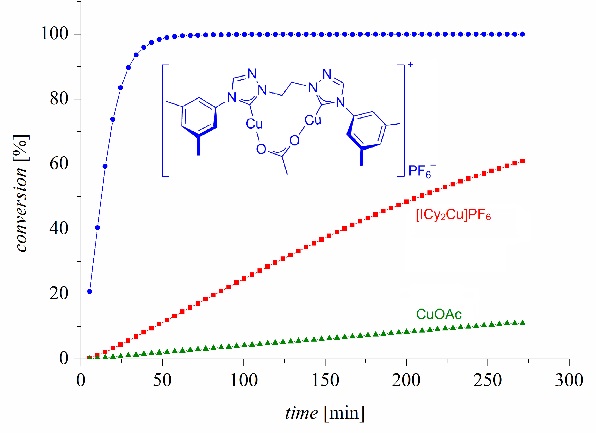
Highly Active Dinuclear Copper Catalysts for Homogeneous Azide-Alkyne-Cycloadditions
Regina Berg, Johannes Straub, Ella Schreiner, Steffen Mader, Frank Rominger, Bernd F. Straub*, Adv. Synth. Catal. 2012, 354, 3445-3450.
Advancements in the mechanistic understanding of the copper-catalyzed azide–alkyne cycloaddition
Regina Berg*, Bernd F. Straub*, Beilstein J. Org. Chem. 2013, 9, 2715-2750.
Our EurJIC issue cover picture  shows a Madagascar day gecko in a coconut with a locust on top. Gecko toes hold on to slithery surfaces due to van der Waals forces, which are also relevant for the lipophilic altebate anion.
shows a Madagascar day gecko in a coconut with a locust on top. Gecko toes hold on to slithery surfaces due to van der Waals forces, which are also relevant for the lipophilic altebate anion.

A Lipophilic, Fluorine-Free, Thermostable, Inexpensive, S4-Symmetric, Highly Soluble, Weakly Coordinating, Protolabile Aluminate
Bernd F. Straub*, Michael Wrede, Karin Schmid, Frank Rominger, Eur. J. Inorg. Chem. 2010, 1907-1911.

